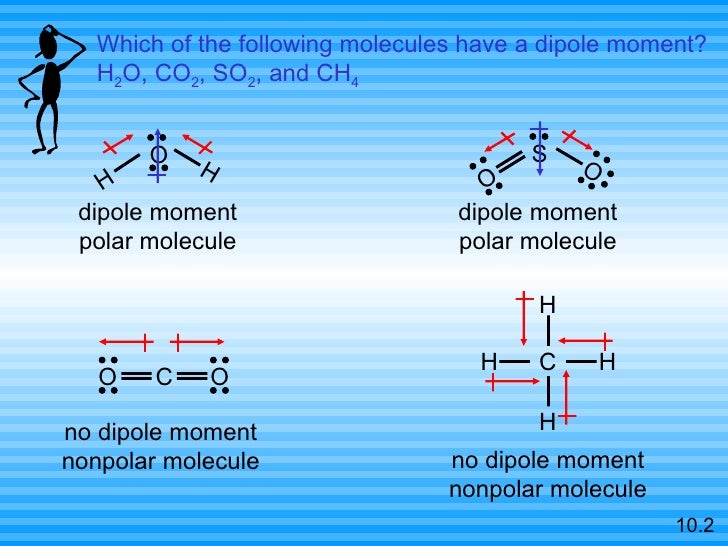
There are better methods, but more costly for the level of the question addressed, however, I thought it already provides the insight necessary here. Sidenote: a force field like MMFF94 is a computationally cheap approach to optimize molecular structures. For diatomic molecules, there is only one bond, so its bond dipole moment determines the molecular polarity. We determine the dipole moment by adding the bond moments in three-dimensional space, taking into account the molecular structure. Hence the molecule of dichloromethane is more polar than the one of chloroform. The dipole moment measures the extent of net charge separation in the molecule as a whole. However, beside the angle, distances like C-Cl and C-H are affected by the different steric demand and electronic properties, too equally influencing the overall dipole moment. Simultaneously, you will recognize the different angle defined by Cl-C-Cl, too. So, C O 2 has zero dipole moment because it is a linear molecule. But, as the dipole moment of one bond is cancelled by other, the structure of C O 2 is a linear. You easily will recognize a feature in common - the similar direction of the resulting dipolar moment. C O 2 has the structure as O C O in which there is bond dipole moment (C-O bond). You are literally able "to walk around" the molecule and its dipolar moment. Lacking access to experimentally determined data about the molecular geometries,you may sketch the molecules in question, for example with Avogadro, and optimize the geometry (here, MMFF94 was deployed). Okay thank you.As the dipolar moment is a vectorial property, you have to take into account both the number of chlorine atoms around the carbon atom, as well as their relative arrangement - among them, as well in respect to the carbon atom. And the direction of men devote moment will be in right direction. That's why this molecule will be polar molecule and new is not zero. They are not cancelled out by each other. There's fire department will be in this direction. So that one moment will be this carbon electron negativity is more than hydrogen. And so at CNN the structure is like that nitrogen is more electric negative than carbon. Okay in that day part of the question we have add to see. So resulting the whole moment will be in above our direction. And the direction of mandible moment will be elected because three double moments in one direction and vulnerable moment is in down over direction. So this molecule is well let them or lead you polar molecule. So you can observe that all the disciples will not cancel out by each other.

Okay so the whole movement direction will be these feds. In the c part of the ocean we have CH2 Cierto. So as to as molecule is a lot of monitor and the nanny type of moment direction will be also a provided direction. Identify the electron pair geometry and the molecular structure of each. So the apple moments will not cancel out because here is long pair also present and the effect of long pair will not cancel out by each other and direction of both, hydrogen is in the same side, so it will not cancelled by each other. For a molecule, the overall dipole moment is determined by both the. Okay, so here that whole moment directions.

And then the molecule we have a choice do s Molecular structure of H two.
#Sf2 dipole moment full
The full moments by each other because the direction is opposite. That's where they will be cancelled loud. F two molecules will be zero and it is non polar molecule florian atoms is the same. Okay so in all these disciples will be cancelled out by each other.
Will we cancel out by each other? Also this is our land type of moment of these of these two diapers. That's why this type of moment will be here and this will be here. Okay so here we can or instead of that that whole movement direction is From less electron negative Adam two hours high electron negativity. Okay so we are going to discuss how can we identify which molecule is polar or non popular in the first molecule we have X E. This intermixing usually results in the formation of hybrid orbitals having entirely different energies, shapes, etc. The question is which of this molecule has a bad type of normal and also indicate the direction of the whole moment in the solitude. Hybridization in Chemistry is defined as the concept of mixing two atomic orbitals to give rise to a new type of hybridized orbitals.
